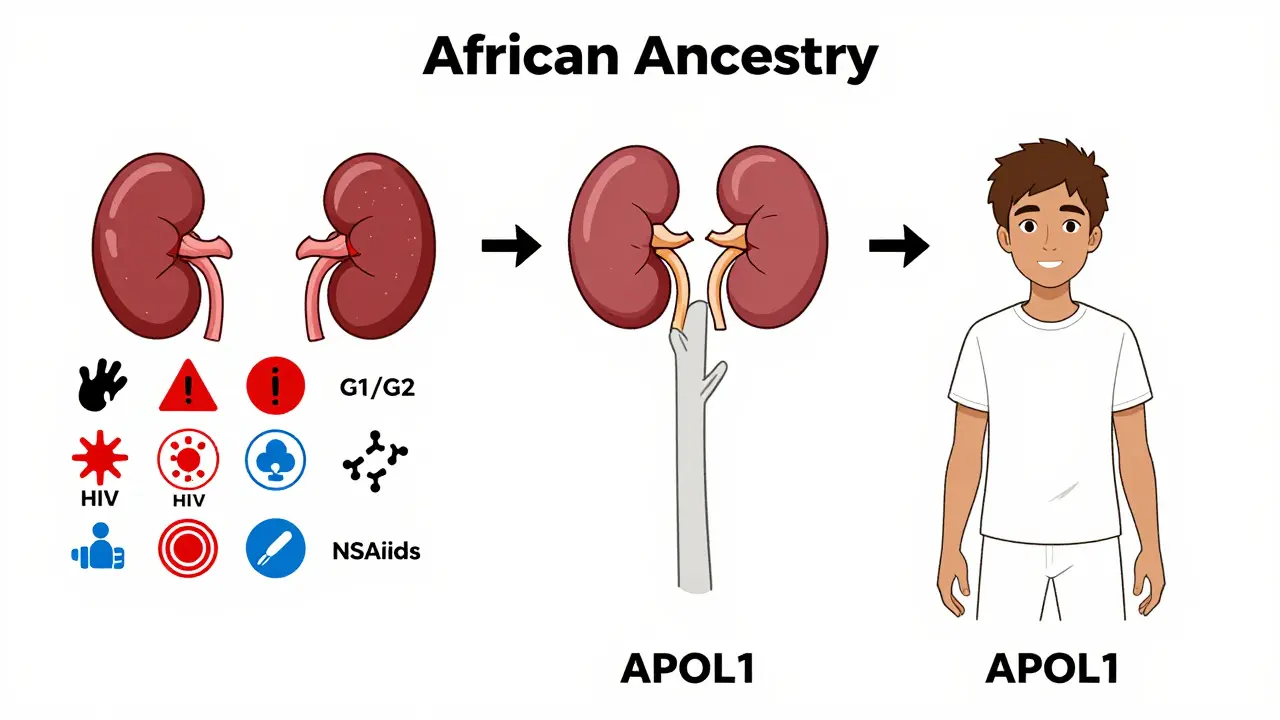
When it comes to kidney disease, not everyone faces the same risk. For people with recent African ancestry, one gene-APOL1-plays a huge role in why kidney failure is so much more common. In fact, this single genetic factor explains about 70% of the extra risk seen in African Americans, Afro-Caribbeans, and others with West African roots. It’s not about race. It’s about ancestry. And understanding APOL1 changes how we think about kidney health, testing, and treatment.
What Is APOL1 and Why Does It Matter?
APOL1 is a gene that makes a protein involved in your body’s immune defense. Long ago, in parts of West and Central Africa, certain changes (called variants) in this gene helped people survive a deadly parasite called Trypanosoma brucei rhodesiense, which causes African sleeping sickness. Those who carried the G1 or G2 versions of APOL1 were more likely to live long enough to have children. Over time, these variants became common in populations from that region.
But here’s the twist: the same changes that saved lives from parasites now put kidney cells at risk. In modern environments, where sleeping sickness is rare, these APOL1 variants can damage the filtering units of the kidney-the glomeruli. This leads to serious conditions like focal segmental glomerulosclerosis (FSGS), collapsing glomerulopathy, and HIV-associated nephropathy (HIVAN).
Who Carries the High-Risk APOL1 Genotype?
Not everyone with African ancestry is at risk. The danger comes only if you inherit two bad copies of the gene-one from each parent. That’s called a high-risk genotype: G1/G1, G2/G2, or G1/G2 (compound heterozygous).
About 13% of African Americans carry this high-risk combination. Among those who already have non-diabetic kidney disease, that number jumps to nearly 50%. In West Africa, around 30% of people in countries like Ghana and Nigeria carry at least one risk variant. But here’s the key point: these variants are almost never found in people of European, Asian, or Indigenous American descent. That’s why the kidney disease gap exists mainly between African ancestry populations and others.
Most People With APOL1 Risk Don’t Get Sick
Here’s something surprising: about 80-85% of people with two risky APOL1 variants never develop kidney disease. That’s called incomplete penetrance. It means having the genotype doesn’t guarantee illness. Something else has to trigger it.
Researchers call these triggers "second hits." They include viral infections like HIV, high blood pressure, obesity, certain medications, or even pregnancy. For example, in the UK, nearly half of all end-stage kidney disease cases in people with HIV and African ancestry were directly tied to APOL1 risk. Without the gene, HIV might not have caused kidney failure. With it? The risk skyrockets.
This is why a 30-year-old with high-risk APOL1 and normal blood pressure might never need dialysis-while a 45-year-old with the same genes, uncontrolled hypertension, and a recent viral infection could lose kidney function in just a few years.
How Is APOL1 Testing Done and Who Should Get It?
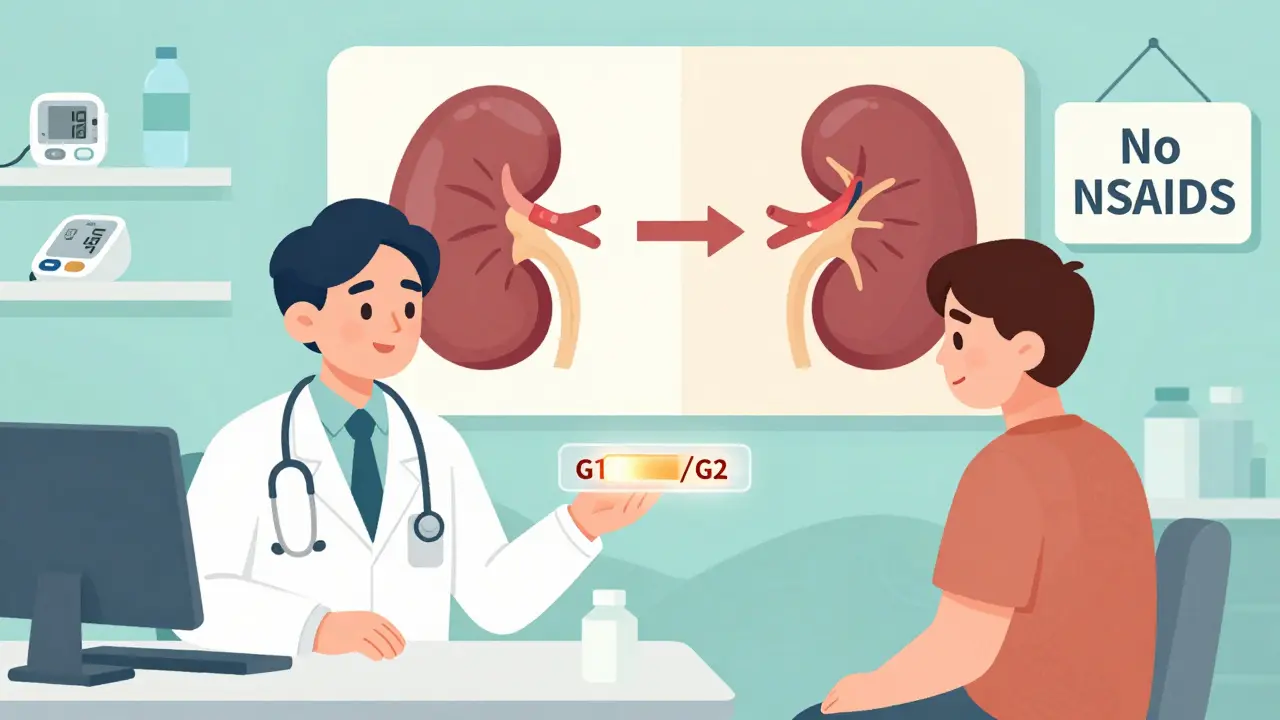
Genetic testing for APOL1 became available in 2016. It’s a simple blood or saliva test that looks for the G1 and G2 variants. Costs range from $250 to $450 without insurance, though some clinical trials and research programs cover it.
The American Society of Nephrology now recommends testing for:
- People of African ancestry with unexplained kidney disease (especially FSGS or collapsing glomerulopathy)
- Living kidney donors with African ancestry (to protect their own long-term kidney health)
- People with HIV and kidney damage
For donors, this isn’t just about the recipient-it’s about the donor’s future. A person with high-risk APOL1 who donates a kidney may have a higher chance of developing kidney failure later in life. Testing helps them make informed decisions.
But here’s the problem: many doctors still don’t know how to interpret the results. A 2022 survey found that 78% of nephrologists felt underprepared to counsel patients. Some patients think a positive test means they’re doomed. Others think it means nothing. The truth? It means monitoring-regular urine tests for protein, blood pressure checks, and avoiding things that stress the kidneys.
Real Stories, Real Impact
One woman, Emani, found out she had two APOL1 risk variants during a routine check-up. She had no symptoms, no family history of kidney disease. But because she knew her risk, she started getting annual urine tests and kept her blood pressure under 120/80. Five years later, her kidneys are still working perfectly.
On the other side, a Reddit user named BlackMedStudent shares how knowing their APOL1 status changed everything. "I check my blood pressure every week. I avoid NSAIDs. I drink water. I don’t smoke. I feel like I’m playing defense for my future kidneys. But I’m also scared every time I get sick-what if this cold becomes a second hit?"
And then there’s the emotional toll. Many patients report being dismissed for years. "My doctor said my protein in urine was just from high blood pressure," one user wrote. "It took five years and a genetic test to realize it was my genes, not just my lifestyle."
What’s Being Done About It?
The science is moving fast. In October 2023, Vertex Pharmaceuticals released Phase 2 results for a drug called VX-147. It blocks the harmful APOL1 protein in the kidney. In a 140-person trial, patients on the drug saw a 37% drop in proteinuria-the main sign of kidney damage-in just 13 weeks. That’s huge.
The NIH has launched a 10-year study called the APOL1 Observational Study (AOS), tracking 5,000 people with high-risk genotypes. They’re trying to figure out what triggers kidney damage and how to stop it.
Meanwhile, the American Society of Nephrology is pushing for:
- Clear screening guidelines by 2026
- Tools to predict who’s most likely to progress by 2025
- Equitable access to testing and future drugs by 2027
And the market is responding. The global APOL1 testing market is expected to grow from $18.7 million in 2022 to over $43 million by 2027. Big pharma is investing billions because this isn’t just a niche condition-it’s a major driver of racial health disparities.
Why This Isn’t About Race
It’s critical to say this clearly: APOL1 risk isn’t about race. It’s about ancestry. Race is a social idea. Ancestry is biology. You can have African ancestry without being Black in a racial sense. And you can be Black without recent African ancestry.
Dr. Olugbenga Gbadegesin put it best: "We must be careful not to conflate social constructs of race with genetic ancestry." Using race to estimate kidney function (like the old race-adjusted eGFR formula) has caused harm. The American Medical Association banned that practice in 2022, partly because of APOL1 research.
APOL1 shows us that the best way to understand health risks is to look at genes-not skin color.
What You Can Do Now
If you have African ancestry and are concerned about kidney health:
- Ask your doctor about an annual urine albumin-to-creatinine ratio test. It’s cheap, non-invasive, and catches early damage.
- Keep your blood pressure under 130/80. Even small spikes can be dangerous if you have high-risk APOL1.
- Avoid NSAIDs like ibuprofen or naproxen unless absolutely necessary-they stress the kidneys.
- If you have HIV, hepatitis, or other chronic infections, talk to your doctor about APOL1 testing.
- Don’t panic if you find out you have the high-risk genotype. Most people never develop disease. But knowledge lets you take control.
And if you’re a healthcare provider: learn the science. Stop using race as a proxy. Test when appropriate. Counsel with care. APOL1 isn’t a death sentence. It’s a warning sign-and warnings are meant to be acted on.
The Bigger Picture
APOL1 is a perfect example of how evolution shapes health in unexpected ways. A mutation that saved lives from a deadly parasite now threatens lives from chronic disease. It’s not a flaw-it’s a consequence of survival.
But science is catching up. We now have tools to identify who’s at risk, ways to monitor them, and drugs in development that could block the damage. The future isn’t about treating kidney disease after it happens. It’s about preventing it before it starts.
For people with African ancestry, that future is closer than ever. And it starts with knowing your genes.





11 Comments
Shourya Tanay
The APOL1 risk variants represent a fascinating case study in evolutionary trade-offs. The G1 and G2 alleles confer resistance to Trypanosoma brucei rhodesiense through a lytic mechanism targeting the parasite's membrane, but in modern contexts, the same protein forms cation channels in podocytes, inducing mitochondrial dysfunction and autophagic cell death. This pleiotropy underscores why population genetics must be decoupled from racial categorization. The penetrance is incomplete because environmental modifiers-like interferon exposure during viral infections-act as second hits, triggering podocyte injury. We need biomarkers for these triggers, not just genotyping.
LiV Beau
This is so important!! 🙌 I had no idea my ancestry could mean my kidneys are ticking time bombs unless I stay on top of BP and avoid ibuprofen. My mom’s a nurse and she’s been nagging me about hydration but now I get why. I just got tested last month-negative for the high-risk combo, but I’m telling all my cousins to get checked. Knowledge is power, y’all!! 💪💧
Adam Kleinberg
So let me get this straight you're saying black people have a gene that makes them more likely to get kidney disease but only if they have two copies and only if they get sick or have high blood pressure and only if they're from west africa but not if they're from east africa and not if they're mixed and not if they're from the caribbean unless they're from jamaica or trinidad but not haiti and not if they're from brazil and not if they're from london and not if they're from canada and not if they're from australia but the drug works and the test costs 300 bucks and the government is pushing it but doctors don't understand it and race is a social construct but we still use it to calculate gfr so what exactly are we supposed to believe here i mean like seriously who wrote this
Gene Forte
Understanding APOL1 is not just a medical breakthrough-it's a moral imperative. Science has given us the tools to move beyond crude racial proxies and toward precision medicine. This isn't about blame or fear. It's about empowerment. Every individual with African ancestry deserves the chance to know their genetic landscape so they can protect their health proactively. We must ensure equitable access to testing, counseling, and emerging therapies. The future of nephrology is personalized-and it starts with awareness.
Kenneth Zieden-Weber
Let’s be real: if this gene were common in white folks, we’d have mandatory screening, national awareness campaigns, and a congressional hearing by now. But because it’s tied to African ancestry? It’s ‘complicated.’ Meanwhile, people are losing kidneys because their doctor didn’t know to test them. The fact that 78% of nephrologists feel unprepared? That’s not ignorance-it’s systemic neglect. And don’t even get me started on how long it took for the race-adjusted eGFR to be ditched. We’re still playing catch-up while people die.
Chris Bird
I am from Nigeria and we have this gene everywhere. My cousin died at 32 from kidney failure. His doctor said he had high BP. But he was fit. He ate well. He never took drugs. Only after his brother got tested did we find out. Now I tell everyone: get tested. It’s free in Lagos at the university hospital. Stop listening to doctors who think you’re just fat or lazy. Your genes are not your fault. But ignoring them? That’s your mistake.
David L. Thomas
The incomplete penetrance is the most fascinating part. We’re seeing a classic gene-environment interaction here-APOL1 risk variants are like loaded guns, but they only fire when the trigger is pulled by a second hit. That means we’re not just looking at genetics, we’re looking at exposomes: viral load, metabolic stress, even psychosocial factors like chronic inflammation from systemic racism. The NIH’s AOS study should be expanded to include longitudinal environmental data. We need to map the triggers, not just the genotype.
Bridgette Pulliam
I work in public health, and I’ve seen firsthand how this information gets buried under layers of mistrust. Many Black communities have been burned by medical research-Tuskegee, Henrietta Lacks, forced sterilizations. So when a new genetic test comes out, the question isn’t 'Is this helpful?'-it’s 'Who benefits?' We need community-led education, not top-down pamphlets. Partner with churches, barbershops, HBCUs. Let trusted voices explain it. And stop using clinical jargon. Say 'your genes make your kidneys more fragile' instead of 'podocyte injury from APOL1-mediated cytotoxicity.'
Mike Winter
It’s striking how the APOL1 story mirrors broader themes in medical genetics: a mutation selected for survival in one context becomes pathogenic in another. This is not a flaw in evolution-it’s a consequence of environmental mismatch. The same mechanism that protected against trypanosomes now destabilizes glomerular filtration under conditions of metabolic stress. The challenge now is not merely to treat, but to predict. We need predictive models integrating genotype, lifestyle, and exposure history. And we must avoid the trap of genetic determinism. Risk is not destiny.
Randall Walker
I have the high-risk genotype. I’m 29. My BP is 118/76. I don’t take NSAIDs. I drink water. I get tested every year. I still wake up every time I get a cold wondering if this is the one. I don’t want kids. I don’t want to risk passing it on. I don’t want to be the one who ends up on dialysis because I didn’t do enough. I’m not scared of the gene. I’m scared of the silence around it.
Miranda Varn-Harper
While I appreciate the scientific rigor of this post, I must note that the emphasis on ancestry as distinct from race is itself a form of performative precision. Genetic ancestry is a statistical construct, not a biological absolute. The notion that 'African ancestry' is a coherent category ignores the vast heterogeneity across the continent. Furthermore, the commercialization of APOL1 testing-now projected to reach $43 million-raises serious ethical questions about who stands to profit from what is essentially a legacy of colonial epidemiology. The real issue is not the gene-it’s the system that only now deigns to care.